UND IRES Research Project
New Green Methodology Using Non-natural Amino Acid Catalysts.
Mentors
Goal
New approaches to unique chiral organocatalysts based on non-natural amino acids and their application as catalysts in selected organic reactions.
Novelty
This project explores a novel approach to a new class of bifunctional organocatalysts based on non-natural polyfunctional amino acids. The scaffold is simple to access and pseudoenantiomeric catalysts are available in few standard steps. Enamine and hydrogen-bonding catalysis are combined in the catalyst design.
Broader Impacts
The skeletal framework of the catalyst can be easily tuned and other catalytic groups can be envisaged to be easily introduced on the scaffold resulting in new catalysis modes and small-molecule organocatalyst generations. Therefore these investigations have multiple implications for future catalysis directions.
Background
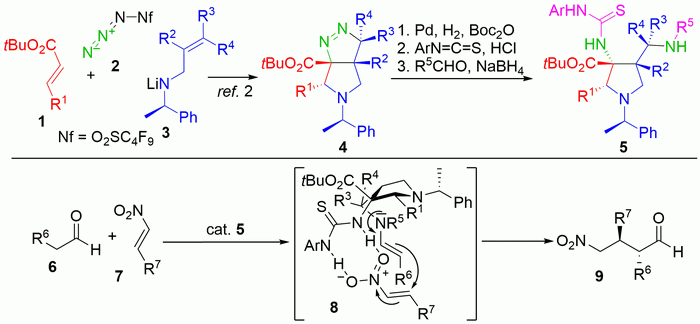
Organocatalysis holds much promise for green synthesis of organic compounds with potential interesting medicinal or materials properties. The use of expensive, toxic and environmentally problematic transition metal catalysts can thus be avoided. Most current organocatalysts are derived from naturally occurring amino acids, which are modified into species that are tailored to interact with the substrates by reversible chemical bonding and/or supramolecular interactions. However, natural amino acids have several disadvantages for catalysis, since the enantiomeric species are not always readily available, and they contain limited functionality to allow tuning of catalytic efficiency. Therefore polyfunctional amino acid-derived catalysts available equally in both enantiomeric forms should be potentially much more valuable. We recently developed a synthesis for such an amino acid in a single step by a new tandem conjugate addition/1,3-dipolar cycloaddition reaction giving new a,b,g-triamino acid derivatives in enantiomerically pure form. Applying them as bifunctional organocatalysts is quite attractive.
Methods
The student will perform the tandem asymmetric amide conjugate addition/diazo transfer/1,3-dipolar cycloaddition reaction between chiral allylamine 1, the alpha,beta-unsaturated ester 2 and stable and safe nonaflyl azide 3 providing access to the bicyclic framework 4 of the projected organocatalysts in one step. This compound will be transformed in three standard steps to the actual catalyst, compound 5. Note that the arrangement of thiourea and amino units can be reversed when Boc2O is omitted in the hydrogenation step. In this way pseudo-enantiomeric catalysts would result from a single precursor. Their catalytic applicability will be initially evaluated in a model Michael addition of aldehyde 6 to nitroalkene 7.